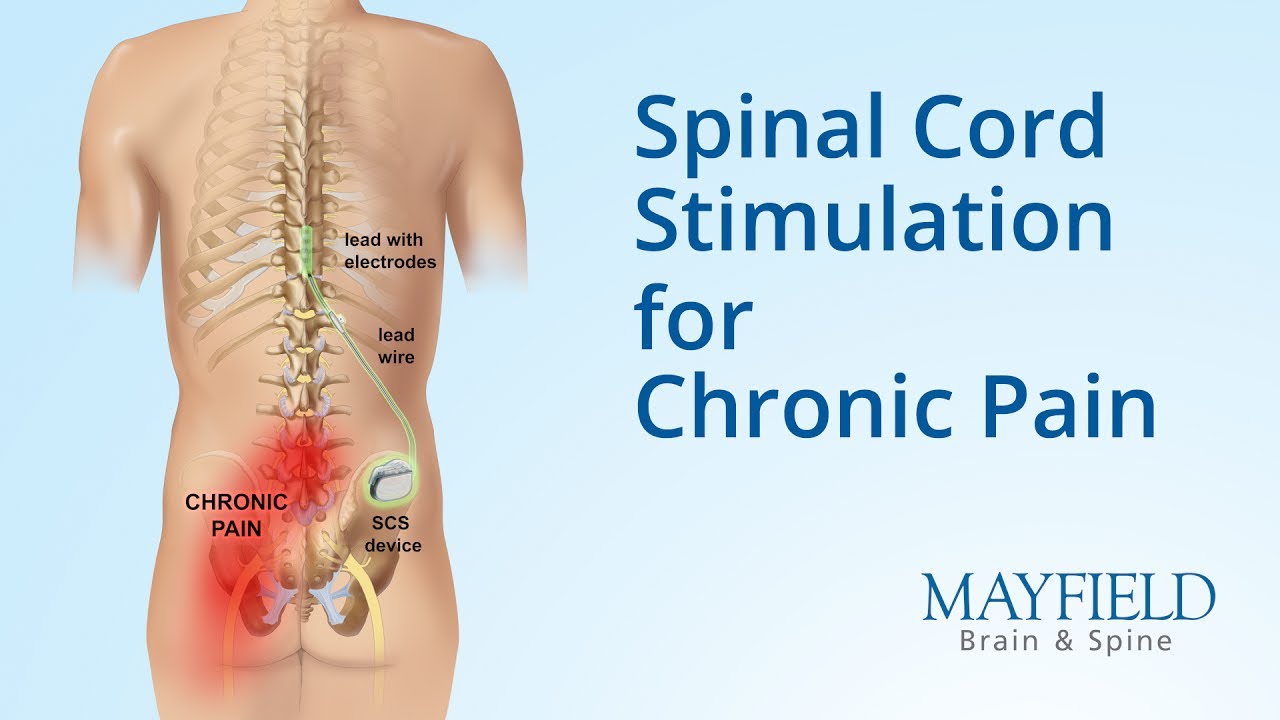
Patients reported an average pain relief of 53%, measured using questionnaires, which was maintained for over 5 years at clinically meaningful levels (≥ 50% pain relief). What will it accomplish?ĭata from published research studies support the effectiveness of Abbott’s Prodigy, Proclaim, and Proclaim XR SCS Systems when treating people with chronic, intractable pain of the trunk and/or limbs caused by diabetic peripheral neuropathy of the lower extremities. When is it used?Ībbott’s Prodigy, Proclaim, and Proclaim XR SCS Systems are used to help manage chronic, intractable pain of the trunk and/or limbs, including one-sided or two-sided pain associated with the following: failed back surgery syndrome, intractable low back and leg pain, and diabetic peripheral neuropathy of the lower extremities. Doctors believe stimulation targets specific nerves that impact how pain is perceived and/or nerve structures that aren’t working properly. The signals tell the IPG when to deliver appropriate stimulation to the spinal cord. The implanted pulse generator (IPG) receives radio signals from the patient controller. This supplement expands the Indications for Use to include treatment of diabetic peripheral neuropathy of the lower extremities through a series of consistent stimulation pulses, called the tonic stimulation mode. The main components of these SCS systems are an implanted pulse (signal) generator that is connected to one or two implanted leads, a patient controller, and a clinician programmer. Product Name: Prodigy, Proclaim, and Proclaim XR Spinal Cord Stimulation (SCS) SystemsĪddress: 6901 Preston Road, Plano, TX 75024 USAĪpproval Letter: Approval Order What is it?Ībbott’s Prodigy, Proclaim, and Proclaim XR Spinal Cord Stimulation (SCS) Systems are implanted spinal cord stimulation systems intended to treat long-term (chronic) pain in the trunk or limbs that is difficult to manage (intractable). See the links below to the Summary of Safety and Effectiveness Data (SSED) and product labeling for more complete information on this product, its indications for use, and the basis for FDA’s approval. This is a brief overview of information related to FDA’s approval to market this product.
